The importance of molecular response in CML
By Professor Susan Branford PhD, a medical scientist with the Department of Genetics and Molecular Pathology, Centre for Cancer Biology, SA Pathology and the University of South Australia and University of Adelaide.
My PhD research in biomedical science involved assessing people with CML to determine if monitoring the level of disease during therapy using a molecular marker could predict treatment outcome.
I also investigated the reasons for drug resistance that occurs in a small number of patients.
The tests I developed are now incorporated into routine patient monitoring along with the other blood tests a person with CML has on a regular basis after diagnosis.
CML is a genetic disease. A change in the structure of DNA within cells leads to the formation of a new gene that does not behave normally and causes CML. The new gene is formed from the fusion of two different genes and is called BCR-ABL1.
BCR-ABL1 is a molecular marker that can be measured during therapy. Importantly, a blood sample is required to measure BCR-ABL1 levels rather than bone marrow, the collection of which is associated with greater discomfort for patients.
In the past, there was a greater need for people with CML to have bone marrow biopsies to diagnose their disease and to monitor their response to therapy. Bone marrow collection is still necessary at diagnosis and is a very important part of patient management.
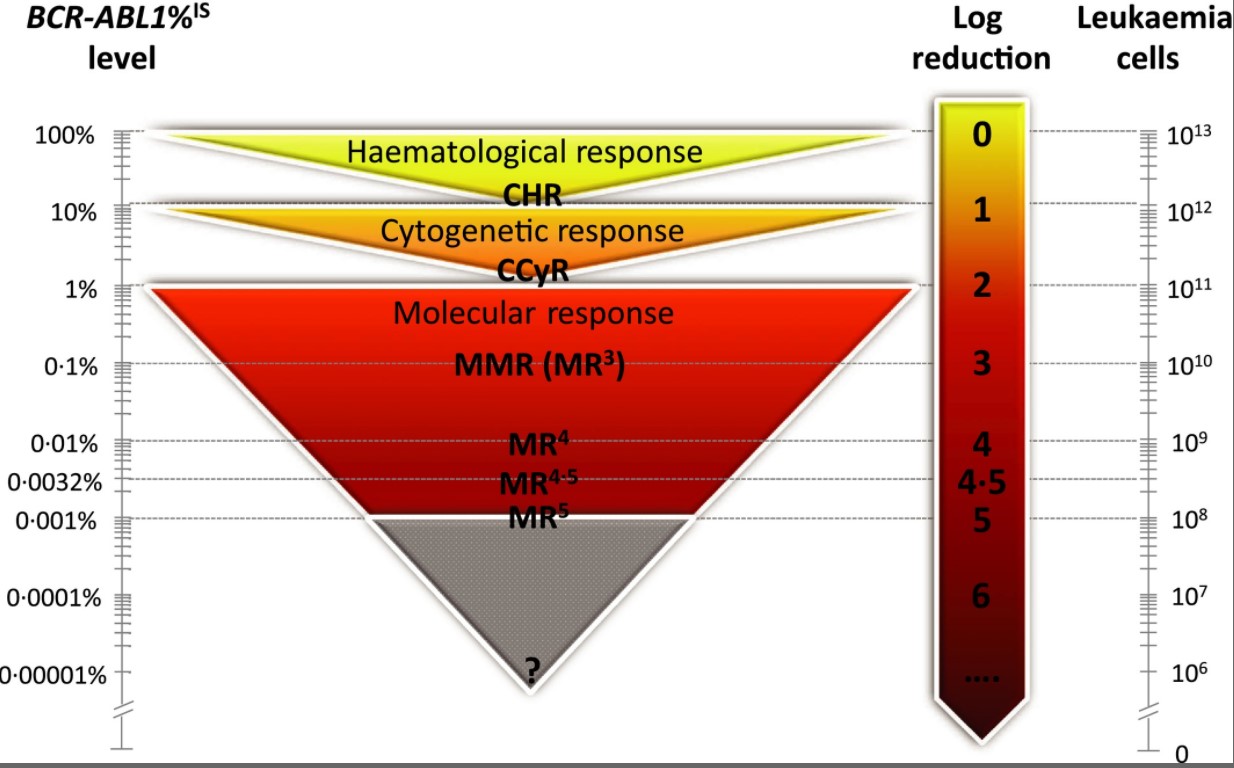
In Australia, however, molecular monitoring of BCR-ABL1 during therapy, using a blood sample, has largely taken over from bone marrow testing. The advantages of molecular testing include a shorter time for the result to be ready, and the test is 300-1000 times more sensitive for detecting low levels of leukaemia, compared with cytogenetic testing.

Our laboratory has been monitoring BCR-ABL1 levels in patients with CML for a long time and was one of the first laboratories in the world to develop this technique.
We monitored patients in early clinical trials for imatinib – the first drug that was truly effective for most CML patients and since then there have been many clinical trials of different and more powerful drugs.
All the trials have consistently shown that the molecular test can provide very useful clinical information for the prediction of response to drug therapy, and many labs around the world are now performing the test.
The molecular technique is not easy for new labs to develop and BCR-ABL1 values can vary from lab to lab. To overcome these differences, about 10 years ago it was decided that all labs adopt a common way to report the results, so a BCR-ABL1 value is similar no matter in which lab it is tested.
Working with many labs around the world, we developed a set of recommendations for the testing procedure and BCR-ABL1 values are now reported on an international reporting scale. This has improved the quality of the results.
A panel of experts recommended that BCR-ABL1 values reported on the international reporting scale be used to determine if a patient is responding well to therapy or if a change of therapy is necessary to improve response.
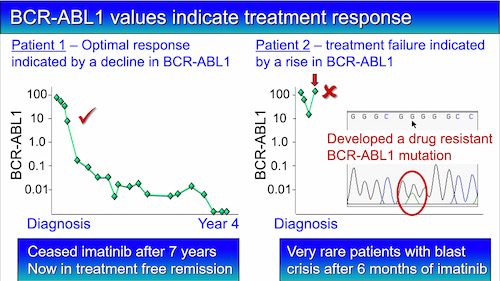
The critical timepoints to assess response is at three, six and 12 months of drug treatment. A value of 10% at three months, 1% at six months, and 0.1% at 12 months is an optimal response, and no change of therapy is necessary. Most patients reach these levels.
A small number of patients develop drug resistance and the main reason is a change in the DNA sequence of the BCR-ABL1 gene that can stop the drug working properly.
A rise in BCR-ABL1 level can indicate that drug resistance is occurring. When a rise occurs and the doctor suspects relapse, a blood sample can be used to check for a change in the DNA sequence of BCR-ABL1. The haematologist can then decide whether a change of therapy is necessary.
There is still a lot we don’t know about CML. At the time of diagnosis, we can’t identify those patients who will fail their therapy. But technology is rapidly advancing, and over the next 10 years, we hope to more thoroughly examine and understand the molecular changes that occur, which will lead to improved treatment and better outcomes for all patients.
Last updated on May 20th, 2021
Developed by the Leukaemia Foundation in consultation with people living with a blood cancer, Leukaemia Foundation support staff, haematology nursing staff and/or Australian clinical haematologists. This content is provided for information purposes only and we urge you to always seek advice from a registered health care professional for diagnosis, treatment and answers to your medical questions, including the suitability of a particular therapy, service, product or treatment in your circumstances. The Leukaemia Foundation shall not bear any liability for any person relying on the materials contained on this website.