New 3D models could revolutionise drug testing
Dr Laura Bray has made it her life’s work to develop 3D models which she hopes will provide answers to the many unknowns faced by people with AML and other life-threatening diseases.

There is so much uncertainty when they are diagnosed, she said. They want to know how aggressive their cancer is, whether it will come back after treatment, and if so where.
“I want to develop predictive technologies that give people more clarity about their illness, personalised treatment, and a better understanding of the long-term risks into the future.”
Dr Bray is a bio-scientist who has worked on three-dimensional (3D) tissue microenvironment models of acute myeloid leukaemia (AML) for seven years.
She is the lead investigator of a new study aimed at improving our understanding of the biology of AML and treatment resistance by developing new models for drug testing.
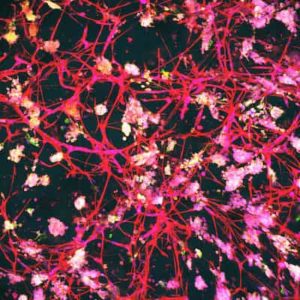
The 3D testing models she is developing more accurately mimic the natural environment of the human bone marrow compared with the traditional two-dimensional (2D) laboratory methods.
Dr Bray is a Group Leader and Deputy Director of the ARC Training Centre for Cell and Tissue Engineering Technologies at the Queensland University of Technology (QUT).

She was awarded a $200,000 Priority-driven Collaborative Cancer Research Scheme grant from the Leukaemia Foundation in collaboration with Cancer Australia (July 2019-July 2022).
The title of Dr Bray’s research project is Development of a translational bioengineered microenvironment model to advance pre-clinical acute myeloid leukaemia research and is supported through the Estate of Florence Brown.
Dr Bray said 3D tissue microenvironments can replicate many natural mechanical, chemical, and cellular processes that cannot be depicted in the 2D cell cultures currently used for most drug research.
“My fascination with 3D models began in 2009, during my PhD project at the Queensland Eye Institute. I was using tissue engineering to grow three-dimensional corneas for potential future applications in patients who needed a replacement,” said Dr Bray.
“I could see the remarkable potential then of these models as an alternative to animal or human testing.”

After being awarded her PhD, Dr Bray moved to Germany for on a three-year research fellowship and her focus shifted to AML.
“I had a real personal drive to bring my knowledge of tissue microenvironments back into the oncology field, as my family, friends and colleagues have been affected by cancer in many different forms,” she said.
“In the lab, when you’re using a standard culture of leukaemia cells, they are just floating around in a solution and we know it’s not replicating the way that leukaemia cells grow in the actual bone marrow environment. There are other cell types in the bone marrow, including blood vessels which act as a conduit to nutrients and flush out waste in the body.
“But these blood vessels can also act to protect leukaemia cells, giving them the ability to hide and become resistant to treatment.
“By developing these 3D bone marrow microenvironments, we hope to understand how these leukaemia cells interact with the surrounding natural cells, and hopefully develop new targets to stop them growing in that space.”
These models have the potential to be utilised as research platforms for fundamental biological studies and pre-clinical trials, as well as to study new targeted therapies for cancer patients.

Dr Bray is eager to see the study build on previous findings which her research group has already published.
“We were able to recreate part of a 3D bone marrow microenvironment and watch the leukaemia cells interact with it in real time,” she said.
“We then applied a certain drug that inhibits the ability of the cancer cells to adhere to the blood vessel and watched the cells successfully detach from the vessels in the model, showing that our models can reproduce quite complex biological processes.”
Having Leukaemia Foundation funding will enable Dr Bray to determine which cells will survive after certain treatments and then start to grow again.
“Over time we need to be able to identify these specific cells and then remove them out of the model to study what makes them resistant,” said Dr Bray.
“By being able to profile these cells, we can hopefully find some new targeted treatments.”
A major driver for developing these models is to reduce the number of animals used for drug testing, and in 2015, Dr Bray was awarded a Lush Prize for her work; the largest global prize fund in the non-animal testing sector.
“The regulatory structure in Australia stipulates that animal testing must be done prior to human trials,” she said.
“But we know that 95% of drugs that have positive results in a lab setting will fail in human clinical trials.
“This will save time, money, and many animals from having to be sacrificed just to find out that the drug will not work on a human.”
Due to the COVID-19 pandemic, Dr Bray’s lab was closed for four months last year and was unable to source specialised blood vessel cells from the U.S. supplier during that time.
“That was really difficult as it takes a lot of time to build up these cell cultures,” explained Dr Bray.
“It takes a few months of work, either side, to wind it down and then start it up again, but hopefully we are through the worst of it now and the Leukaemia Foundation has been really understanding and permitted an extension to my grant funding period.”
Over the past few years, Dr Bray and her team have worked to promote the benefits of 3D models and encourage biologists at the bench to adopt the technology.
“Traditional 2D cultures are easy and they’re cheap, but we and others are trying to bring these 3D models into the mainstream research space.
“This project will certainly help our case with the dream to see these three-dimensional technologies applied across all diseases, not just blood cancer.”
This research project is kindly supported through The Bourne Foundation.
Last updated on March 25th, 2021
Developed by the Leukaemia Foundation in consultation with people living with a blood cancer, Leukaemia Foundation support staff, haematology nursing staff and/or Australian clinical haematologists. This content is provided for information purposes only and we urge you to always seek advice from a registered health care professional for diagnosis, treatment and answers to your medical questions, including the suitability of a particular therapy, service, product or treatment in your circumstances. The Leukaemia Foundation shall not bear any liability for any person relying on the materials contained on this website.