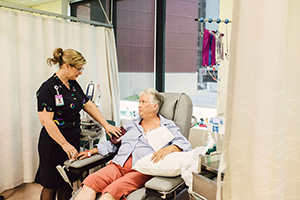
Blood cancer information
Clear, reliable information for people living with blood cancer and their family members, friends, and health professionals.
There are over 120 different types of blood cancer. The Leukaemia Foundation supports people impacted by all blood cancers. Find out the blood cancers and disorders the Leukaemia Foundation supports.
Here you find information on all types of blood cancers (including leukaemia/leukemia, lymphoma and myeloma), as well as information relevant to the blood cancer journey from diagnosis, to treatment, and beyond. There’s also information for carers and friends, and healthcare professionals.
Related information
Awareness activities